Definition of Amylase
Amylase is an enzyme that breaks down the glycosidic bonds between glucose units to catalyse the hydrolysis of starch to create smaller sugar units.
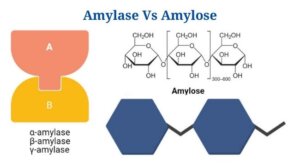
- There are two varieties of amylases: α-amylase as well as β-amylase, that hydrolyze the α-as well as β – linkages in the starch molecule, respectively.
- In addition to α – as well as β-amylases, living systems include a third category of amylases called γ -amylases, that cleave the glycosidic bonds present at the non-reducing end of amylose as well as amylopectin.
- Amylose as well as amylopectin, that are connected together by α-1,4- glycosidic as well as α -1,6-glycosidic connections, make up starch.
- Amylase is a protein made up of peptide bonds that connect a number of amino acid units.
- Since it is classified as a tertiary protein, it has a complex structure that includes hydrogen bonds, hydrophobic bonds, disulfide bonds, as well as ionic bonds.
- Amylases are glycoside hydrolases that break down the glycosidic link in polymeric sugar molecules.
- Of all the amylases found in the human body, α-amylases are the most abundant. Different glands in the body, such as the salivary glands and the pancreas, create these amylases.
- Amylases are created as carbs by other animals, microorganisms, as well as plants that are the major source of energy for all living things.
- Amylases break down polysaccharides into disaccharides as well astrisaccharides that are subsequently broken down further by other enzymes into glucose as well as other monosaccharides to supply energy for metabolism.
- Despite the fact that amylases’ primary function in living systems is digestion, they are also required for industrial application in the food as well as beverage sectors.
- In the fermentation of starch-rich cereals to make alcoholic beverages, both – as well as -amylases are involved.
- Amylase is a flour addition that can be used in breadmaking as well as other bakery procedures. These are also used in medicine to break down bigger saccharides into simple sugars as part of pancreatic enzyme replacement therapy.
Definition of Amylose
Amylose is a polysaccharide made up of α -D-glucose units connected together by α(1→4) glycosidic linkages.
- Amylose is a structure found in polysaccharides such as starch that is more resistant to digestion than other structures of the same type.
- The amount of glucose units in amylose typically ranges from 300 to 3000, but in exceptional situations, the number can reach tens of thousands.
- Amylose is a polysaccharide with glycosidic linkages connecting the monomeric units (glucose or other monosaccharides).
- Amylose is particularly significant in starch since of its helical structure, that makes it resistant to digestion. Aside from that, the linear structure of the molecules takes up less space. As a result, plants can store amylose for later use.
- Although the structure of amylose is less complex than that of amylase, it can exist in disordered amorphous or helical forms.
- Amylose makes up roughly 20–25 percent of total starch volume, with the balance being made up of amylopectin, a distinct type of polysaccharide.
- Amylose has a straight chain structure with no branches. As a result, the molecule only has a α-1,4- glycosidic linkage. Amylases break down these connections during digestion, allowing monosaccharides to be digested during metabolism.
- Amylose comprises α-D- glucose units that are formed when the carbon 1 of one glucose molecule is linked to the carbon-4 of the other.
- Amylases degrade amylose into maltotriose as well as maltose that can then be digested further to generate energy.
- Amylose may be detected in laboratories using an iodine solution that produces a dark blue-colored solution showing amylose presence.
- These can also be utilised in food as well as textile products as thickeners, water binders, as well as stabilisers.
Key Differences Between Amylase and Amylose
(Amylase vs Amylose)
Amylase and Amylose Citations
- https://byjus.com/chemistry/amylose/
- http://www.differencebetween.net/science/health/difference-between-salivary-and-pancreatic-amylase/
- https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/amylase
- https://www.difference.wiki/amylose-vs-amylopectin/
- https://www.askdifference.com/starch-vs-polysaccharide/
Related Posts
- Dissecting Microscope (Stereo Microscope) Definition, Principle, Uses, Parts
- Saturated vs Unsaturated Hydrocarbons: Definition, Differences, Examples
- Ethanol Vs Methanol: Definition and 10+ Differences
- Hydrogen Bond: Properties, Definition, Types, Examples
- Nitrate Vs Nitrite: Definition, Differences, Examples
- Aromatic Compounds vs Aliphatic Compounds: Definition, Differences, Examples
- Compound Vs Mixture: Definition, Differences, Examples
- Elements Vs Compounds: Definition, Differences, Examples
- Molecules Vs Compounds: Definition, Differences, Examples
- Hard water Vs Soft water: Definition, Differences, Examples
- Glucose Vs Sucrose: Definition and Key Differences
- 13+ Difference Between Atom and Molecule with Examples
- How to Balance Chemical Equation: Methods, Steps, Examples
- Ionic Bond: Definition, Properties, Examples
- Amylase Vs Amylose: Definition, Differences, Example