Definition of Ethanol
Ethanol, often known as ethyl alcohol, is the primary alcohol produced by replacing a hydrogen atom in the alkane ethane with a hydroxyl group.
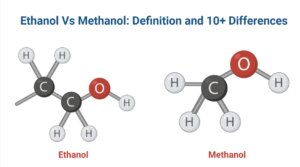
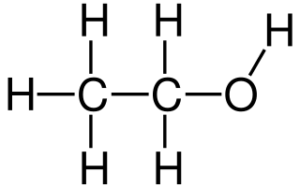
Fig: Ethanol Structure, Ethanol Formula C2H5OH
- It has the chemical formula C2H5OH as well as is an organic molecule made up of carbon, hydrogen, as well as oxygen. CH3-CH2-OH is the chemical formula.
- It’s a highly flammable, highly volatile liquid with a mild odor. It’s frequently used as a psychotropic or recreational drug.
- Ethanol can be made spontaneously by yeast as well as other microbes fermenting carbohydrates. It can also be made synthetically via petrochemical methods such as ethylene hydration.
- Ethanol is one of the most well-known alcohols, as well as many people are familiar with it as the active ingredient in alcoholic beverages.
- It is also utilized as a chemical solvent in the synthesis of several other organic compounds as well as is considered a potential fuel source.
- Ethyl alcohol can be used as an antiseptic or disinfectant in addition to being utilized in alcoholic beverages.
- Ethanol is also utilized in lightweight rocket-powered racing aircraft as a rocket fuel.
- Ethanol is less acidic than water since it is less polar than methanol. The presence of two methyl groups allows electrons to be distributed across multiple atoms, lowering the molecule’s polarity.
Definition of Methanol
Methanol is the main alcohol generated when a hydrogen atom in the alkane methane is substationed by a hydroxyl group.
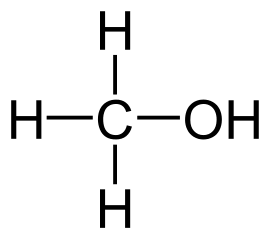
Fig: Methanol Structure, Methanol Formula CH3OH
- Methanol is a highly flammable as well as volatile liquid with a distinctive alcoholic odor comparable to that of ethanol.
- Methanol was previously known as wood alcohol since it was made by distilling wood. It is now predominantly made via the hydrogenation of carbon monoxide.
- Methanol has the chemical formula CH3OH, where the methyl group is joined to a polar hydroxyl group.
- Methanol is the most basic aliphatic alcohol as well as is frequently utilized as an amphiprotic solvent, fuel, as well as human metabolite.
- Methanol vapors can entirely combine with water, but they are slightly heavier than air as well as can only travel a short distance to a source of ignition.
- Methanol is an extremely poisonous alcohol that, in small doses, can result in lifelong blindness.
- Methanol toxicity can manifest itself in two ways: it can be converted into formic acids that are poisonous since they inhibit mitochondrial cytochrome c oxidase, or it can induce deadly effects on the central nervous system in the form of depressants.
- Methanol poisoning epidemics have been reported as a result of methanol contamination of drinking alcohol.
Image Created with BioRender
Key Differences between Ethanol and Methanol
(Ethanol and Methanol)
[ninja_tables id=”5697″]
Ethanol and Methanol Citations
- National Center for Biotechnology Information. “PubChem Compound Summary for CID 702, Ethanol” PubChem, https://pubchem.ncbi.nlm.nih.gov/compound/Ethanol. Accessed 16 March 2021.
- https://www.uchemchemicals.com/organic-intermediates/organic-reagent-and-other-heterocycles/methyl-alcohol-cas-67-56-1.html
- https://www.differencebetween.com/difference-between-ethanol-and-vs-methanol/
- https://en.wikipedia.org/wiki/Anhydrous_ethanol
- https://www.vedantu.com/chemistry/methanol
- https://www.osti.gov/biblio/5260923-catalytic-hydrogenation-carbon-monoxide
- https://www.hazmattool.com/info.php?a=Methanol&b=UN1230&c=3
- https://www.foodstandards.gov.au/publications/documents/TR2.pdf
- https://www.engineeringtoolbox.com/ethanol-ethyl-alcohol-properties-C2H6O-d_2027.html
- https://www.differencebetween.com/difference-between-ethanol-and-bioethanol/
- https://www.chemicalsafetyfacts.org/ethanol/
- https://petrochemistry.eu/interactive-flowchart/
- https://pediaa.com/difference-between-ethyl-alcohol-and-ethanol/
- https://pediaa.com/difference-between-ethanol-and-methanol/
- https://conservative.ly/toxic-methanol-that-causes-blindness-found-in-hand-sanitizers-fda-warns
- https://brainly.com/question/11006244
Related Posts
- Dissecting Microscope (Stereo Microscope) Definition, Principle, Uses, Parts
- Saturated vs Unsaturated Hydrocarbons: Definition, Differences, Examples
- Ethanol Vs Methanol: Definition and 10+ Differences
- Hydrogen Bond: Properties, Definition, Types, Examples
- Nitrate Vs Nitrite: Definition, Differences, Examples
- Aromatic Compounds vs Aliphatic Compounds: Definition, Differences, Examples
- Compound Vs Mixture: Definition, Differences, Examples
- Elements Vs Compounds: Definition, Differences, Examples
- Molecules Vs Compounds: Definition, Differences, Examples
- Hard water Vs Soft water: Definition, Differences, Examples
- Glucose Vs Sucrose: Definition and Key Differences
- 13+ Difference Between Atom and Molecule with Examples
- How to Balance Chemical Equation: Methods, Steps, Examples
- Ionic Bond: Definition, Properties, Examples
- Amylase Vs Amylose: Definition, Differences, Example