Definition of Amylose
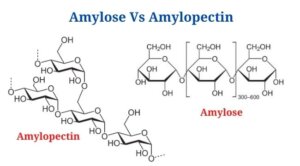
Amylose is a polysaccharide made up of α-D-glucose units connected together by α(1→4) glycosidic linkages.
- Amylose is a type of resistant starch that can withstand digestion better than other starch molecules. The molecule’s resistance is aided by its closely packed helical shape.
- In glucose, the carbon atoms are numbered, starting with carbon 1 at aldehyde, that is linked to the 4-carbon of the following molecule, resulting in an α(1→4) glycosidic connection.
- Amylose contains between 300 – 3000 glucose units, however the number can reach thousands in extreme situations.
- Amylose chains can take one of three forms: disordered amorphous conformation, helical conformation, or both.
- The chain can form a double helix with itself or with other molecules like iodine as well as fatty acids.
- Amylose’s lengthy chains crystallise more easily than other types of glucose, making it more resistant to heat as well as digestive enzymes.
- Amylose is found in starch at a concentration of 20-25 percent, where glucose molecules are compacted into long chains.
- A higher concentration of amylose in starch reduces the starch‘s expansion potential as well as gel strength.
- Amylose is a component of starch molecules, hence it’s abundant in plants. Meyer as well as his coworkers identified it during their maize research in 1940.
- It’s important for plant storage since it’s less digestible than amylopectin as well as takes up less space due to its helical form.
- The digestive enzyme -amylase breaks down the chains into maltotrioseas well as maltose.
- Amylose can be utilised in food as well as industry as a thickener, water binder, as well as emulsion stabiliser.
- Iodine can be used to detect amylose that results in a dark blue-black colour.
Definition of Amylopectin
Amylopectin is a polysaccharide that is made up of a branching polymer of α-glucose units.
- Amylopectin’s glucose units are linked in a linear chain by α(1→4)glycosidic bonds, with branching occurring between 20 as well as 30 glucose units by α(1→6) glycosidic linkages.
- In the amylopectin molecule, α(1→6)bonds account for around 4-6 percent of overall connections. Three different sorts of chains make up the molecule.
- The A chains are linear chains with a single α(1→6) linkage, whereas the B chains are chains connected by α(1→6) linkages to other chains. The centre linear chains, or C chains, are made up of the reduction group (aldehyde group).
- Amylopectin is a component of starch that makes about 80-85 percent of the total weight, but the concentration varies depending on the source.
- Despite the fact that amylopectin makes up 80 percent of the starch in most plants, waxy starches contain 100 percent amylopectin.
- The crystalline domains of the starch granules are formed by the clustered branches of amylopectin chains.
- The ratio of amylopectin to amylose affects the functional properties of the starch-like viscosity, solubility, texture, as well as gel stability.
- Amylopectin molecules are significantly larger than amylose molecules, as well as they have a clustered branched structure.
- Instead of hard clumps, these molecules frequently form soft pastes of gels that are characterised by clarity, stability, as well as ageing resistance.
- Despite the fact that amylopectin is the most abundant component of starch, there is no reliable way for measuring it directly, as well as most amylopectin research relies on enzymatic as well as instrumental procedures.
- These approaches have revealed that diverse plant sources of starch have varying branch chain lengths as well as amylopectin dispersion.
Key Difference between Amylose and Amylopectin
(Amylose Vs Amylopectin)
Amylose and Amylopectin Citations
- https://www.difference.wiki/amylose-vs-amylopectin/
- https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/amylopectin
- https://www.sciencedirect.com/science/article/pii/S0268005X12002974
- https://www.sciencedirect.com/topics/engineering/iodine-number
- https://www.sciencedirect.com/topics/engineering/amylopectin
- https://www.sciencedirect.com/science/article/pii/S0144861719301857
- https://www.researchgate.net/publication/275662036_Determination_of_amyloseamylopectin_ratio_of_starches
Related Posts
- Dissecting Microscope (Stereo Microscope) Definition, Principle, Uses, Parts
- Saturated vs Unsaturated Hydrocarbons: Definition, Differences, Examples
- Ethanol Vs Methanol: Definition and 10+ Differences
- Hydrogen Bond: Properties, Definition, Types, Examples
- Nitrate Vs Nitrite: Definition, Differences, Examples
- Aromatic Compounds vs Aliphatic Compounds: Definition, Differences, Examples
- Compound Vs Mixture: Definition, Differences, Examples
- Elements Vs Compounds: Definition, Differences, Examples
- Molecules Vs Compounds: Definition, Differences, Examples
- Hard water Vs Soft water: Definition, Differences, Examples
- Glucose Vs Sucrose: Definition and Key Differences
- 13+ Difference Between Atom and Molecule with Examples
- How to Balance Chemical Equation: Methods, Steps, Examples
- Ionic Bond: Definition, Properties, Examples
- Amylase Vs Amylose: Definition, Differences, Example