Aerobic Respiration
Glycolysis (Cytoplasm
Krebs’s cycle (Mitochondria)
Electron transport chain & oxidative phosphorylation
- It involves complete oxidation of P.A. by removing H Atom, leaving three molecules of CO2, and passing electron removed as part of H-atom to molecular O2 in ETS for ATP synthesis.
- The first process takes place in the matrix of mitochondria and the second in the inner membrane.
- Pyruvate undergoes oxidase decarboxylation by pyruvic dehydrogenase in a matrix including NAD+ and Co-A.
- PA + CoA + NAD+ → Acetyl Co-A + CO2 +NADH + H+ (in presence of Mg2+ & Pyruvate dehydrogenase).
- 2 molecule NADH is produced from 2 molecules of P.A.
- Acetyl Co- A enters TCA- Krebs’s Cycle (by Trans Krebs)
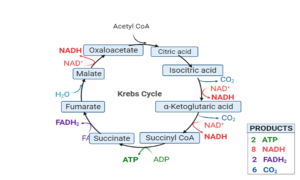
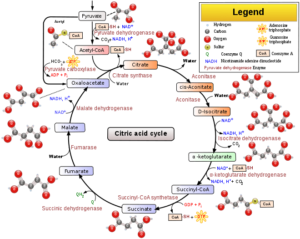
Krebs Cycle or Tricarboxylic Acid Cycle or TCA Cycle
Where does the citric acid cycle occur?
Location: Krebs cycle occurs in the mitochondrial matrix
First product is citric acid.
The citric acid cycle (CAC) – also known as the TCA cycle (tricarboxylic acid cycle) or the Krebs cycle – is a series of chemical reactions to release stored energy through the oxidation of acetyl-CoA derived from carbohydrates, fats, and proteins. The TCA cycle is used by organisms that respire (as opposed to organisms that ferment) to generate energy, either by anaerobic respiration or aerobic respiration. In addition, the cycle provides precursors of certain amino acids, as well as the reducing agent NADH, that are used in numerous other reactions. Its central importance to many biochemical pathways suggests that it was one of the earliest components of metabolism and may have originated a biogenically. Even though it is branded as a ‘cycle’, it is not necessary for metabolites to follow only one specific route; at least three segments of the citric acid cycle have been recognized.
Click Here for All Biology Notes
Steps in the Krebs Cycle or Citric Acid Cycle
It is an eight-step process. Krebs cycle or Citric Acid Cycle takes place in the matrix of mitochondria under aerobic condition.
Step 1. The first step is a condensation step, combining the two-carbon acetyl group (from acetyl CoA) with a four-carbon oxaloacetate molecule to form a six-carbon molecule of citrate. CoA is bound to a sulfhydryl group (-SH) and diffuses away to eventually combine with another acetyl group. This step is irreversible because it is highly exergonic. The rate of this reaction is controlled by negative feedback and the amount of ATP available. If ATP levels increase, the rate of this reaction decreases. If ATP is in short supply, the rate increases.
Step 2. Citrate loses one water molecule and gains another as citrate is converted into its isomer, isocitrate.
Steps 3 and 4. In step three, isocitrate is oxidized, producing a five-carbon molecule, α-ketoglutarate, together with a molecule of CO2 and two electrons, which reduce NAD+ to NADH. This step is also regulated by negative feedback from ATP and NADH and by a positive effect of ADP. Steps three and four are both oxidation and decarboxylation steps, which release electrons that reduce NAD+ to NADH and release carboxyl groups that form CO2 molecules. α-Ketoglutarate is the product of step three, and a succinyl group is the product of step four. CoA binds the succinyl group to form succinyl CoA. The enzyme that catalyzes step four is regulated by feedback inhibition of ATP, succinyl CoA, and NADH.
Step 5. A phosphate group is substituted for coenzyme A, and a high- energy bond is formed. This energy is used in substrate-level phosphorylation (during the conversion of the succinyl group to succinate) to form either guanine triphosphate (GTP) or ATP. There are two forms of the enzyme, called isoenzymes, for this step, depending upon the type of animal tissue in which they are found. One form is found in tissues that use large amounts of ATP, such as heart and skeletal muscle. This form produces ATP. The second form of the enzyme is found in tissues that have a high number of anabolic pathways, such as liver. This form produces GTP. GTP is energetically equivalent to ATP; however, its use is more restricted. In particular, protein synthesis primarily uses GTP.
Step 6. Step six is a dehydration process that converts succinate into fumarate with the help of an enzyme called succinate dehydrogenate. Two hydrogen atoms are transferred to FAD, producing FADH2. The energy contained in the electrons of these atoms is insufficient to reduce NAD+ but adequate to reduce FAD. Unlike NADH, this carrier remains attached to the enzyme and transfers the electrons to the electron transport chain directly. This process is made possible by the localization of the enzyme catalyzing this step inside the inner membrane of the mitochondrion.
Step 7. Water is added to fumarate during step seven, and malate is produced. The last step in the citric acid cycle regenerates oxaloacetate by oxidizing malate. Another molecule of NADH is produced.
Krebs Cycle Products /TCA Cycle Product
Each citric acid cycle forms the following products:
- 2 molecules of CO2 are released. Removal of CO2 or decarboxylation of citric acid takes place at two places:
- In the conversion of isocitrate (6C) to 𝝰-ketoglutarate (5C)
- In the conversion of 𝝰-ketoglutarate (5C) to succinyl CoA (4C)
- 1 ATP is produced in the conversion of succinyl CoA to succinate
- 3 NAD+ are reduced to NADH and 1 FAD+ is converted to FADH2 in the following reactions:
- Isocitrate to 𝝰-ketoglutarate → NADH
- 𝝰-ketoglutarate to succinyl CoA → NADH
- Succinate to fumarate → FADH2
- Malate to Oxaloacetate → NADH
Note that 2 molecules of Acetyl CoA are produced from oxidative decarboxylation of 2 pyruvates so two cycles are required per glucose molecule.
Krebs Cycle Equation
To Sum up
Significance of Krebs Cycle
-
Krebs cycle or Citric acid cycle is the final pathway of oxidation of glucose, fats and amino acids.
-
Many animals are dependent on nutrients other than glucose as an energy source.
-
Amino acids (metabolic product of proteins) are deaminated and get converted to pyruvate and other intermediates of the Krebs cycle. They enter the cycle and get metabolised e.g. alanine is converted to pyruvate, glutamate to α-ketoglutarate, aspartate to oxaloacetate on deamination.
-
Fatty acids undergo β-oxidation to form acetyl CoA, which enters the Krebs cycle.
-
It is the major source of ATP production in the cells. A large amount of energy is produced after complete oxidation of nutrients.
-
It plays an important role in gluconeogenesis and lipogenesis and interconversion of amino acids.
-
Many intermediate compounds are used in the synthesis of amino acids, nucleotides, cytochromes and chlorophylls, etc.
-
Vitamins play an important role in the citric acid cycle. Riboflavin, niacin, thiamin and pantothenic acid as a part of various enzymes cofactors (FAD, NAD) and coenzyme A.
-
Regulation of Krebs cycle depends on the supply of NAD+ and utilization of ATP in physical and chemical work.
-
The genetic defects of the Krebs cycle enzymes are associated with neural damage.
-
As most of the processes occur in the liver to a significant extent, damage to liver cells has a lot of repercussions. Hyperammonemia occurs in liver diseases and leads to convulsions and coma. This is due to reduced ATP generation as a result of the withdrawal of α-ketoglutarate and formation of glutamate, which forms glutamine.
Electron- transport system (ETS) and oxidative phosphorylation.
- In inner mitochondrial membrane.
- Pathway through which electron passes from one carrier to another.
- It serves 3 essential functions.
- Regenerates oxidized form of Coenzyme to be used in glycolysis & Krebs’s cycle.
- Transports 2H+ and 2 electrons to O2 and utilized the energy of Coenzyme in the production of ATP.
- Electrons from NADH + H+ are oxidized by NADH dehydrogenase & transported through complex 1 to ubiquinone complex – inner membrane and protons are moved from matrix to 1 H.S. electron from FADH2 are transferred from complex II through ubiquinone and protons are moves from matrix to 1 H.S.
- Ubiquinone transferase to complex III and then to complex IV through cyt. C. there, some protons are moved from matrix to intermembrane space.
- Cyt. ‘c’ is a mobile carrier attached to the inner membrane and helps in the transfer of an electron between complex III & IV, complex IV contains cyt. ‘a’, 2 cu center, and cytoplasm a3. It transfers an electron to the final acceptor O2.
- O2 on receiving electron react with 2 H+ reduces to H2O.
2H+ + 2e- + ½ O2 → H2O + energy
- Electron transport and H+ movements create a proton gradient.
- Proton from the intermembrane needs to be pumped to the matrix, but the mitochondrial membrane is impermeable for the proton. So, they are pumped through ATP synthase (complex v). It has Fo, & F.
- F0- integral protein; channel for protein transport.
- F1- Peripheral; catalytic site for ATP synthesis.
- Energy derived from proton pumping is used for ATP synthesis.
2 H+ pumped → 1 ATP synthesis.
NADH→ 3 mol. ATP, 1 FADH2→ 2 mol. ATP; 1 GTP→ 1 mol. ATP.
- It uses energy conserved in Coenzyme during redox reaction to create H+ gradient for phosphorylation, unlike photophosphorylation where the light is utilized – oxidative phosphorylation.
- Total ATP = Glycolysis + Krebs cycle +ETS.
= 4 ATP +10 NADH2 + 2FADH2
= 4 + (10×3) + 2(2) = 33 ATP.
Respiratory Balance sheet –
- NADH in glycolysis is transferred to mitochondria undergoes oxidative phosphorylation.
- None of the intermediates is used to synthesis another compound.
- Only glucose is being respired. No other alternative substrate is entering the pathway at any stage.
Amphibolic pathway –
Catabolism – Breakdown of complex substances to give energy.
Anabolism – Synthesis of complex substances from simple.
Plants also use proteins and fats as respiratory substrates.
Fats→ Glycerol → PGAL
→ Fatty acids → Acetyl Co A
Shows R.P. catabolism.
Protein (in the presence of protease) → Amino acids → Pyruvate
Anabolism:
- PEP (formed during glycolysis is withdrawn) is used to synthesize of carbohydrates & proteins.
- Plant withdraws acetyl CoA and Citrate for the synthesis of fatty acids.
- So, R.P. is involved in both catabolism and anabolism – amphibolia.
Related Posts
- Lineweaver–Burk Plot with Example
- Cell Wall: Definition, Diagram, Structure And Functions
- Phylum Porifera: Classification, Characteristics, Examples
- Dissecting Microscope (Stereo Microscope) Definition, Principle, Uses, Parts
- Epithelial Tissue Vs Connective Tissue: Definition, 16+ Differences, Examples
- 29+ Differences Between Arteries and Veins
- 31+ Differences Between DNA and RNA (DNA vs RNA)
- Eukaryotic Cells: Definition, Parts, Structure, Examples
- Centrifugal Force: Definition, Principle, Formula, Examples
- Asexual Vs Sexual Reproduction: Overview, 18+ Differences, Examples
- Glandular Epithelium: Location, Structure, Functions, Examples
- 25+ Differences between Invertebrates and Vertebrates
- Cilia and Flagella: Definition, Structure, Functions and Diagram
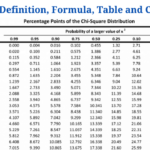
- P-value: Definition, Formula, Table and Calculation
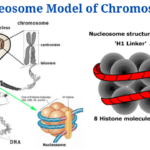
- Nucleosome Model of Chromosome