Fermentation Definition
“Fermentation is an anaerobic process in which energy can be released from glucose even if oxygen is not available.”
What is Fermentation?
Fermentation is the breaking down of sugar molecules into simpler compounds to produce substances that can be used in making chemical energy. Chemical energy, typically in the form of ATP, is important as it drives various biological processes. Fermentation does not use oxygen; thus, it is “anaerobic”
- In yeast, muscle during exercise.
- Incomplete oxidation of glucose. (Absence of O2).
- P.A. → Ethanol +CO2 by pyruvic acid decarboxylase & alcohol dehydrogenase.
- In muscles during exercise, P.A. is reduced to lactic acid by lactate dehydrogenase. Reducing agent NADH + H+, which is deoxidized to NAD+.
- Not much energy is released (less than 7% energy in glucose is released).
- Yeast poison themselves when concentration of alcohol reaches about 13; Hazardous in nature.
Types of Fermentation
There are three different types of fermentation:
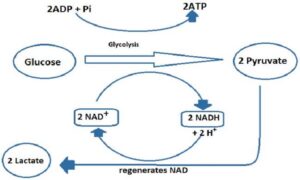
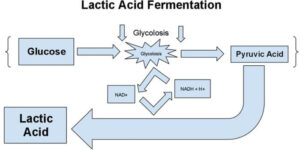
Lactic acid Fermentation
In this, starch or sugar is converted into lactic acid by yeast strains and bacteria. During exercise, energy expenditure is faster than the oxygen supplied to the muscle cells. This results in the formation of lactic acid and painful muscles.
- Curd has a sour taste due to the formation of lactic acid by fermentation by lactobacillus.
- In muscle cells, lactate dehydrogenase connects pyruvate to lactate; leads to stiffness in muscles.
Click Here for Complete Biology Notes
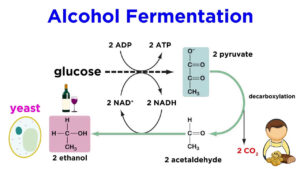
Alcoholic Fermentation
Another familiar fermentation process is alcohol fermentation, in which NADH donates its electrons to a derivative of pyruvate, producing ethanol.
Going from pyruvate to ethanol is a two-step process. In the first step, a carboxyl group is removed from pyruvate and released in as carbon dioxide, producing a two-carbon molecule called acetaldehyde. In the second step, NADH passes its electrons to acetaldehyde, NAD+ and forming ethanol.
Alcohol fermentation by yeast produces the ethanol found in alcoholic drinks like beer and wine. However, alcohol is toxic to yeasts in large quantities (just as it is to humans), which puts an upper limit on the percentage alcohol in these drinks. Ethanol tolerance of yeast ranges from about 555 percent to 212121 percent, depending on the yeast strain and environmental conditions.
Acetic Acid Fermentation
Starch and sugar present in grains and fruits ferment into vinegar and condiments. E.g. apple cider vinegar
Related Posts
- Lineweaver–Burk Plot with Example
- Cell Wall: Definition, Diagram, Structure And Functions
- Phylum Porifera: Classification, Characteristics, Examples
- Dissecting Microscope (Stereo Microscope) Definition, Principle, Uses, Parts
- Epithelial Tissue Vs Connective Tissue: Definition, 16+ Differences, Examples
- 29+ Differences Between Arteries and Veins
- 31+ Differences Between DNA and RNA (DNA vs RNA)
- Eukaryotic Cells: Definition, Parts, Structure, Examples
- Centrifugal Force: Definition, Principle, Formula, Examples
- Asexual Vs Sexual Reproduction: Overview, 18+ Differences, Examples
- Glandular Epithelium: Location, Structure, Functions, Examples
- 25+ Differences between Invertebrates and Vertebrates
- Cilia and Flagella: Definition, Structure, Functions and Diagram
- P-value: Definition, Formula, Table and Calculation
- Nucleosome Model of Chromosome